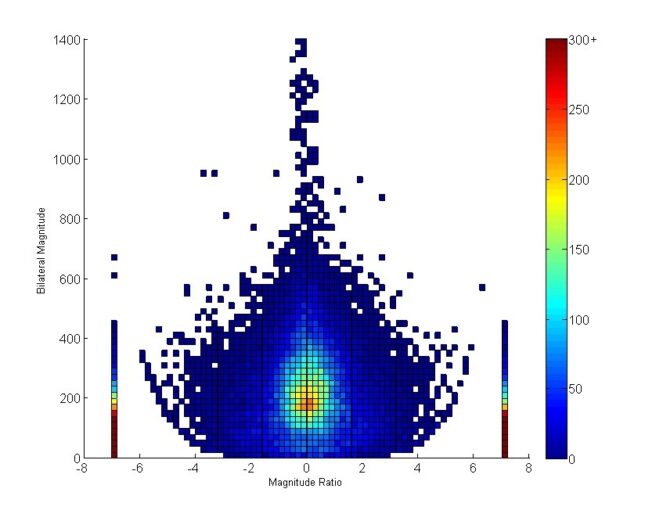
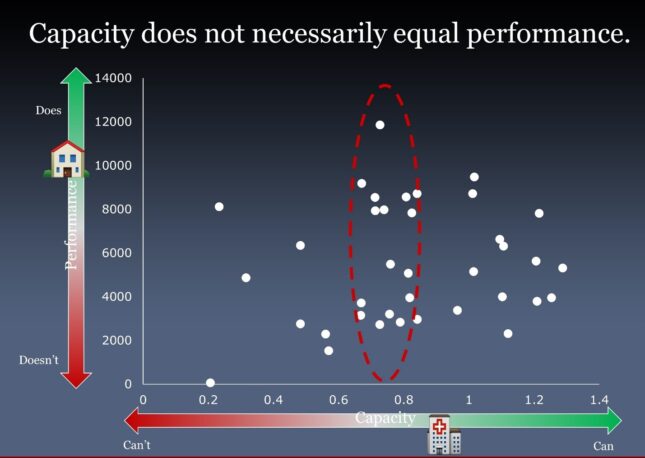
Our research is aimed at developing effective and efficient, individualized rehabilitation for people with neurological and neurodevelopmental conditions. A key tool for our lab is the use of wearable sensors (accelerometers) to quantifying human movement in daily life. This tool offers a convenient, accurate, and economical way of measuring real-world movement outside the clinic or laboratory.
Our laboratory studies the neural control of movement and motor learning in humans. We are interested in better understanding neural control so that this knowledge can be leveraged to inform diagnoses and selection of rehabilitation interventions for persons with stroke, autism, and other neurological conditions. Our ultimate goal is personalized, effective, and efficient interventions that will improve daily life. Since 2004, we have deployed a variety of research tools, including: computerized analyses of human movement (kinematics, kinetics, electromyography) to quantify behavior during ecologically-valid laboratory tasks; standardized clinical assessments to quantify the capacity to produce skilled movement; biosensors (accelerometers) that quantify types and amount of movements outside the laboratory; and standardized questionnaires that quantify self-perception of function, behavior, and participation in everyday life. Ongoing partnerships with others add additional approaches (fMRI, TMS, serum marker analyses) to the available tool set. The interdisciplinary, interactive laboratory environment promotes collaborations within and outside the lab with scientists and clinicians, resulting in a productive, successful research program.
Faculty Investigators
Catherine Lang, PT, PhD, FAPTA [Profile ![]() ]
]
Marghuretta D. Bland, PT, DPT, NCS [Profile ![]() ]
]
Carey L Holleran, PT, MPT, DHS, NCS [Profile ![]() ]
]
Trainees and Staff Members
Allison Miller, DPT, PhD, NCS, Post Doctoral Fellow
Chelsea Macpherson, PT, DPT, PhD, NCS, Post Doctoral Fellow
Christine Gordon, Research PT
McKenna Dixon, DPT Student
Brandon Jensen, DPT Student
Holden Siena, DPT Student
Current Research Studies
Translation of in-clinic gains to gains in daily life
Funding source: NIH R01HD068290 (PI: Lang)
A key purpose of providing rehabilitation services post stroke is to improve performance in daily life. It has long been assumed that improvements in capacity, where capacity is defined as what a person is capable of doing in the structured environment of the clinic or laboratory, result in improvements in real-world performance, where performance is defined as what a person actually does in daily life, outside of the clinic or laboratory. This project uses wearable sensor methodology (accelerometers) to investigate discrepancies between functional capacity and daily performance of activity. We address the following questions:
- Is this discrepancy unique to the upper limb post stroke, or a broader problem across populations and areas of rehabilitation?
- When in the time course of recovery does upper limb performance stabilize?
- How do stroke survivor attitudes and barriers interact with upper limb performance over time?
Variation in early motor function in autism, cerebellar injury, and normal twins
Funding source: NIH R01MH123723 (Co-PIs: Lang, Limperopoulis, Marrus)
A major barrier to improved outcomes in autism spectrum disorder is the challenge of diagnosis early in life. This project deploys wearable sensor methodology in three cohorts between the ages of 6 and 36 months of age to identify and validate two critical motor phenotypes (neural liabilities) contributing to the development of autism: hyperactivity and motor incoordination. These phenotypes result in high risk of developing autism, but cannot yet be reliably measured within the first year of life.
Collaborative Research Studies
- Telerehabilitation in the home after stroke: A randomized, controlled, assessor-blind, clinical trial (TR-2 Trial).
- Funding source:NIH UG3 NS133283 (Co-I; Co-PIs: Cramer and Edwards)
- This definitive STROKENET clinical trial will evaluate the efficacy of home-based telerehabilitation for improving upper limb function, overall health, and economic costs in persons post stroke. Dr. Lang and her team lead efforts for the wearable movement sensor aspects of this study.
Past Research Studies
- Development of a Micro-ECoG Neuroprosthesis for Motor Rehabilitation in a Chronic Corticospinal Stroke Injury, (NIH R01NS101013, (PIs: Moran and Leuthardt))
- Ischemic conditioning as a neurorecovery agent for stroke (NIH R01HD085930)
- Characterizing Arm Recovery in People with Severe Stroke (CARPSS) CIHR/IRSC 374601 (PI: Boyd)
- A Brain Recovery Core for measuring the effectiveness of stroke care (Barnes Jewish Hospital Foundation, WashU McDonnell Center for Systems Neuroscience)
- Enhanced Medical Rehabilitation of older adults
NIH R01MH099011 (PI: Lenze) - Harnessing neuroplasticity to enhance functional recovery in allogenic hand transplant and heterotopic hand replant recipients
DoD W81XWH-15-2-0037 (PI: Frey) - Spinal control during functional activities to improve low back pain outcomes
NIH R01HD047709 (PI: Van Dillen) - Effects of movement context on hemiparetic grasping after stroke (NIH R01HD055964)
- Mechanisms underlying loss of hand function after stroke (NIH K01HD047669)